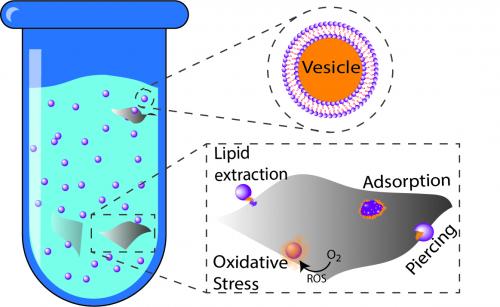
While engineered nanomaterials have known toxic effects, the precise mechanisms of toxicity are not fully understood. In this publication, Ines Zucker, from the Elimelech lab, and her team of collaborators have taken further steps toward understanding how engineered nanomaterials interact with biological materials. This work on vesicle breakage by nanosheets has just been published in Environmental Science & Technology Letters.
Loss of Phospholipid Membrane Integrity Induced by Two-Dimensional Nanomaterials
I Zucker, JR Werber, ZS Fishman, SM Hashmi, UR Gabinet, X Lu, CO Osuji, LD Pfefferle, M Elimelech
DOI: 10.1021/acs.estlett.7b00358
Abstract:
The interaction of two-dimensional (2D) nanomaterials with biological membranes has important implications for ecotoxicity and human health. In this study, we use a dye-leakage assay to quantitatively assess the disruption of a model phospholipid bilayer membrane (i.e., lipid vesicles) by five emerging 2D nanomaterials: graphene oxide (GO), reduced graphene oxide (rGO), molybdenum disulfide (MoS2), copper oxide (CuO), and iron oxide (α-Fe2O3). Leakage of dye from the vesicle inner solution, which indicates loss of membrane integrity, was observed for GO, rGO, and MoS2 nanosheets but not for CuO and α-Fe2O3, implying that 2D morphology by itself is not sufficient to cause loss of membrane integrity. Mixing GO and rGO with lipid vesicles induced aggregation, whereas enhanced stability (dispersion) was observed with MoS2 nanosheets, suggesting different aggregation mechanisms for the 2D nanomaterials upon interaction with lipid bilayers. No loss of membrane integrity was observed under strong oxidative conditions, indicating that nanosheet-driven membrane disruption stemmed from a physical mechanism rather than chemical oxidation. For GO, the most disruptive nanomaterial, we show that the extent of membrane integrity loss was dependent on total surface area, not edge length, which is consistent with a lipid-extraction mechanism and inconsistent with a piercing mechanism.